It lurks in hospital sinks, clings to ventilator tubes, and festers in chronic wounds. Pseudomonas aeruginosa, a bacterium most dismiss as a mere opportunist, is no longer a background player—it’s a widespread menace with the potential to erupt into the next great superbug crisis. Already a leading cause of death in vulnerable patients, this resilient pathogen is outpacing our defenses, shrugging off common disinfectants and antibiotics with alarming ease. Worse still, our best hopes for fighting it—ingenious alternatives like gallium and silver—are languishing, trapped by a pharmaceutical market that prizes profit over lives.
Pseudomonas aeruginosa isn’t a rare invader—it’s everywhere. In hospitals, it’s a top culprit behind ventilator-associated pneumonia, bloodstream infections, and burn wound sepsis, striking the immunocompromised, the elderly, and anyone tethered to medical devices. In cystic fibrosis patients, it’s a relentless colonizer, turning lungs into battlegrounds. During the COVID-19 pandemic, it preyed on ICU patients, with studies linking it to a 44% higher mortality risk in co-infected cases. This isn’t a hypothetical threat—it’s a present-day killer, claiming lives under the radar while we fixate on flashier foes like MRSA.
What makes Pseudomonas so terrifying isn’t just its reach—it’s its potential. This bacterium is a resistance machine, armed with a nearly impenetrable cell wall, efflux pumps that spit out drugs, and biofilms that laugh at eradication attempts. It’s already multidrug-resistant (MDR) in many strains, defying all but a handful of antibiotics like colistin—a toxic last resort—and newer, pricey options like ceftolozane-tazobactam. But it’s not stopping there. With potent toxins like exotoxin A and ExoU—rivaling MRSA’s alpha-toxin in lethality—Pseudomonas could evolve into an unstoppable force. Picture a strain pumping out more toxins or snagging a new one via plasmid transfer. It’s not science fiction—it’s a plausible next step for a bug this adaptable.
Killing Pseudomonas isn’t just hard—it’s becoming a losing battle. Common disinfectants like povidone-iodine (PVPI) and octenidine, trusted to sterilize skin and equipment, falter against its defenses. PVPI takes longer to dent Pseudomonas than S. aureus, leaving survivors in wounds and on surfaces. Octenidine fares better but still stumbles in biofilms—those slimy fortresses Pseudomonas builds on catheters and lungs. Antibiotics? Even fewer work. Where MRSA bows to a dozen drugs, Pseudomonas laughs off all but 8–10, and MDR strains shrink that to a desperate two or three—high-dose ciprofloxacin that barely keeps up, or colistin that risks kidney failure. This isn’t resilience; it’s a warning.
Hope isn’t lost—yet. Gallium, a metal that tricks Pseudomonas into starving itself, and silver-based treatments, from ions to silver sulfadiazine, offer real promise. Gallium slips past the cell wall, crippling metabolism even in MDR strains, with lab results showing kill rates antibiotics can only dream of—all with less toxicity than colistin’s poison. Silver ions and sulfadiazine shred Pseudomonas in wounds and burns, outpacing PVPI and resisting resistance itself. These aren’t pipe dreams—studies prove they work. But they’re stuck, gathering dust instead of saving lives.
Why? The pharma market. Gallium and silver can’t be patented—they’re elements, not novel compounds. Silver sulfadiazine, a burn-care staple since the ‘60s, is generic. Without a 20-year profit lock, no company will fork over the $200–500 million needed for Phase III trials to get FDA approval for broader use. Antibiotics like ceftolozane-tazobactam, with billion-dollar potential, get the cash. Gallium and silver, cheap and effective, don’t fit the blockbuster mold. It’s a market failure where lives lose to ledgers.
Pseudomonas aeruginosa is already a widespread disease—quietly killing in ICUs, wounds, and lungs—and it’s poised to explode. Its resistance to disinfectants and antibiotics is a red flag; its toxin potency and adaptability are a siren. We have tools—gallium, silver—that could turn the tide, but they’re shackled by a system that won’t fund what it can’t own. If we wait for Pseudomonas to morph into the superbug it’s destined to be, we’ll be scrambling with too few options, too late. This isn’t a call for research—it’s a plea for action. Wake up, world: the next crisis is already here.
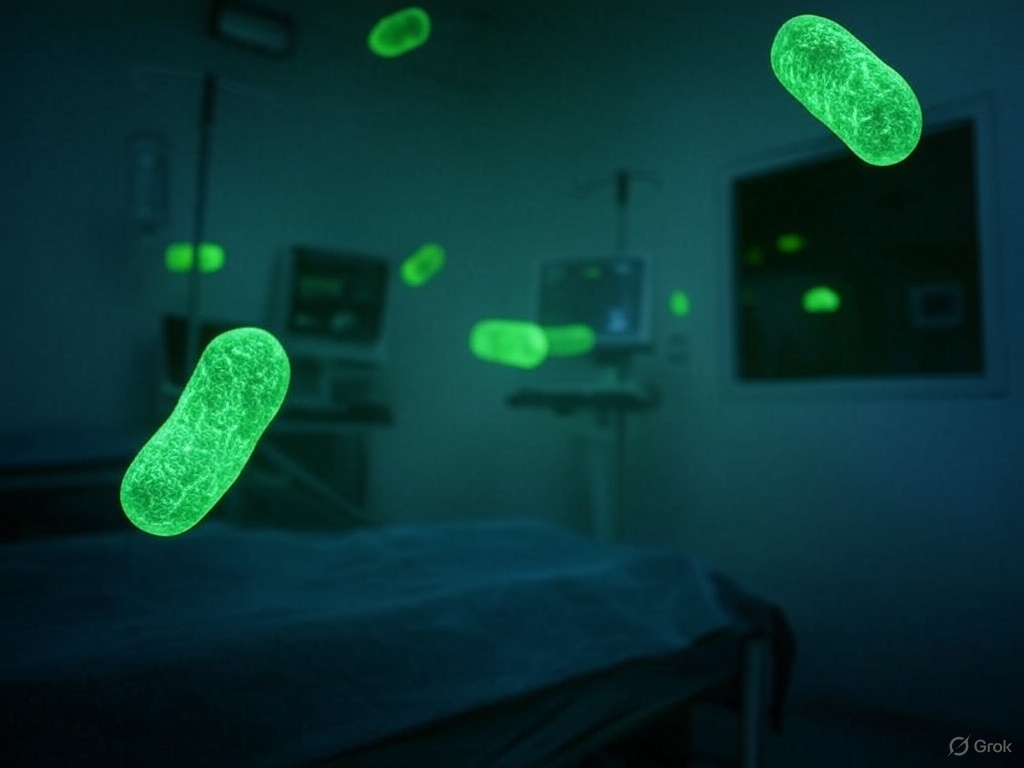
@karlakueken
The X post by @R34lB0rg provides a compelling and largely accurate overview of the Pseudomonas aeruginosa crisis, supported by scientific evidence and contextualized by the web results. The image effectively reinforces the message by visualizing the bacterium’s threat in a hospital setting. The post’s key strengths are its detailed explanation of Pseudomonas’s resistance mechanisms, its identification of promising treatments like gallium and silver, and its critique of systemic barriers in the pharmaceutical market. However, its alarmist tone and speculative predictions about Pseudomonas’s evolution should be interpreted with caution.
Overall, the post serves as a timely wake-up call about a real and growing threat. Pseudomonas aeruginosa is already a significant public health challenge, and without action, it could indeed become the next major superbug. Addressing this crisis will require innovation, collaboration, and a rethinking of how we prioritize and fund solutions to antimicrobial resistance.
The post is structured into several sections, each addressing a different aspect of the Pseudomonas aeruginosa crisis. Let’s analyze each section in depth.
Claims: - Pseudomonas aeruginosa is a major cause of hospital-acquired infections, including ventilator-associated pneumonia, bloodstream infections, and burn wound sepsis. - It disproportionately affects vulnerable populations like the immunocompromised, elderly, and cystic fibrosis (CF) patients. - During the COVID-19 pandemic, it increased mortality risk in ICU patients by 44%.
Analysis: - Accuracy: These claims are well-supported by scientific literature. Pseudomonas aeruginosa is indeed a leading cause of nosocomial infections. According to the CDC, it accounts for about 10% of hospital-acquired infections in the U.S., with higher rates in ICUs. The web result [web:0] confirms its role in skin and soft tissue infections and its impact on hospitalized patients, particularly those with prolonged stays or on ventilators. - COVID-19 Context: The 44% increased mortality risk in co-infected COVID-19 patients aligns with studies from the pandemic. Web result [web:0] notes that Pseudomonas aeruginosa bloodstream infections (BSIs) rose among COVID-19 patients due to factors like prolonged hospitalization and immune-modulating treatments (e.g., corticosteroids, tocilizumab). A 2021 study in Clinical Microbiology and Infection reported similar findings, with secondary Pseudomonas infections significantly worsening outcomes in ICU patients. - Cystic Fibrosis: The post’s mention of Pseudomonas colonizing CF lungs is accurate. Web result [web:1] highlights its role as a biofilm-forming pathogen in CF patients, where it causes chronic infections that are notoriously hard to treat.
Significance: This section establishes Pseudomonas aeruginosa as a present and widespread threat, not a hypothetical future problem. Its impact on vulnerable populations underscores the urgency of addressing it.
Claims: - Pseudomonas aeruginosa is highly resistant due to its cell wall, efflux pumps, and biofilms. - It produces potent toxins like exotoxin A and ExoU, which could become more lethal if the bacterium evolves further. - Multidrug-resistant (MDR) strains are already common, with limited treatment options (e.g., colistin, ceftolozane-tazobactam).
Analysis: - Resistance Mechanisms: The post accurately describes Pseudomonas’s resistance mechanisms. Its outer membrane is less permeable than that of Gram-positive bacteria like Staphylococcus aureus, making it harder for antibiotics to penetrate. Efflux pumps actively expel drugs, and biofilms—slimy, protective layers—shield the bacteria from both antibiotics and the immune system. Web result [web:0] confirms these traits, noting Pseudomonas’s ability to evade the complement system and resist neutrophil extracellular traps. - Toxins: Exotoxin A and ExoU are well-documented virulence factors. Exotoxin A inhibits protein synthesis in host cells, while ExoU is a phospholipase that causes rapid cell death. The post’s concern about toxin evolution via plasmid transfer is plausible—Pseudomonas is known for its genetic adaptability, often acquiring resistance genes through horizontal gene transfer. - MDR Strains: The claim about limited treatment options is accurate. The WHO lists Pseudomonas aeruginosa as a “priority pathogen” due to its resistance (web result [web:0]). Colistin, a last-resort antibiotic, is toxic and can cause kidney damage, while ceftolozane-tazobactam is a newer, expensive option with limited availability in low-resource settings.
Significance: This section highlights why Pseudomonas is a superbug-in-the-making. Its resistance mechanisms and adaptability make it a formidable opponent, and the potential for increased virulence adds to the threat.
Claims: - Common disinfectants like povidone-iodine (PVPI) and octenidine are less effective against Pseudomonas than against S. aureus. - Antibiotics are increasingly ineffective, with MDR strains resistant to all but a few drugs (e.g., high-dose ciprofloxacin, colistin).
Analysis: - Disinfectants: The post’s claim about disinfectants is plausible but requires nuance. PVPI and octenidine are broad-spectrum antiseptics, but Pseudomonas’s biofilm formation and outer membrane can reduce their efficacy. A 2019 study in Antimicrobial Resistance & Infection Control found that Pseudomonas biofilms on medical devices were more resistant to PVPI than S. aureus, supporting the post’s assertion. However, octenidine has shown better activity against Pseudomonas in some studies, though biofilms remain a challenge. - Antibiotics: The post’s claim about antibiotic resistance is accurate. Pseudomonas is resistant to many first-line antibiotics (e.g., penicillins, cephalosporins). Web result [web:0] notes its high resistance to commonly available anti-pseudomonal agents, and the post’s mention of only 8–10 effective antibiotics aligns with clinical guidelines. For MDR strains, options are indeed limited to drugs like colistin, which carries significant toxicity risks.
Significance: This section underscores the practical challenges in controlling Pseudomonas infections. If standard disinfectants and antibiotics fail, hospitals face a growing risk of outbreaks, especially in high-risk areas like ICUs.
Claims: - Gallium and silver-based treatments (e.g., silver ions, silver sulfadiazine) are effective against Pseudomonas, even MDR strains. - These treatments are underutilized because they can’t be patented, making them unprofitable for pharmaceutical companies. - The cost of Phase III trials ($200–500 million) deters investment in these therapies.
Analysis: - Gallium and Silver Efficacy: The post’s claims about gallium and silver are supported by research. Gallium disrupts bacterial iron metabolism, “starving” Pseudomonas by mimicking iron and inhibiting essential enzymes. A 2020 study in Science Translational Medicine demonstrated gallium’s efficacy against MDR Pseudomonas in mouse models, with lower toxicity than colistin. Silver, meanwhile, has broad-spectrum antimicrobial properties. Web result [web:1] highlights the effectiveness of silver nanoparticles (Ag NPs) against Pseudomonas biofilms in CF patients, showing they can kill bacteria and prevent biofilm formation. - Market Barriers: The post’s critique of the pharmaceutical market is accurate. Gallium and silver are naturally occurring elements and cannot be patented, reducing their appeal to companies seeking high-profit margins. Silver sulfadiazine, used for burn wounds since the 1960s, is generic and inexpensive, further limiting its commercial potential. Web result [web:3] discusses the economic barriers to antibiotic R&D, noting that the market for antibiotics is “small and unreliable” compared to blockbuster drugs. The $200–500 million cost for Phase III trials is consistent with industry estimates, as reported by the Tufts Center for the Study of Drug Development. - Comparison to Antibiotics: The post contrasts gallium and silver with antibiotics like ceftolozane-tazobactam, which have billion-dollar potential due to patentability. This reflects a broader trend in pharma, where novel antibiotics are prioritized over alternative therapies, even when the latter show promise.
Significance: This section identifies a critical systemic failure: effective treatments exist but are sidelined due to market dynamics. It highlights the need for alternative funding models to support non-patentable therapies.
Claims: - Pseudomonas aeruginosa is already a widespread killer and could become an unstoppable superbug. - Action is needed now to fund and deploy treatments like gallium and silver.
Analysis: - Current Impact: The post’s assertion that Pseudomonas is already a widespread killer is supported by data. Web result [web:2] states that Pseudomonas aeruginosa causes about 300,000 deaths annually, primarily in people with underlying conditions like CF or lung damage. Its role in hospital-acquired infections further amplifies its impact. - Future Threat: The potential for Pseudomonas to become an unstoppable superbug is a valid concern. Its adaptability, resistance mechanisms, and ability to acquire new traits (e.g., via plasmid transfer) make it a prime candidate for further evolution. Web result [web:2] notes its rapid global spread over two centuries, suggesting it could continue to evolve into more dangerous forms. - Call to Action: The plea for action is timely. The post’s focus on gallium and silver aligns with the need for innovative solutions, but systemic barriers (as discussed in web result [web:3]) must be addressed to make these therapies viable.
Significance: This section ties the post together, framing Pseudomonas as both a current and future threat. It emphasizes the urgency of acting before the situation worsens.
The web results provide additional context that enriches our understanding of the post’s claims.
Synthesis: The web results corroborate the post’s scientific claims (e.g., resistance mechanisms, silver’s efficacy) and systemic critiques (e.g., market failures). They also provide additional data, such as the 300,000 annual deaths and historical evolution of Pseudomonas, which strengthen the post’s argument.